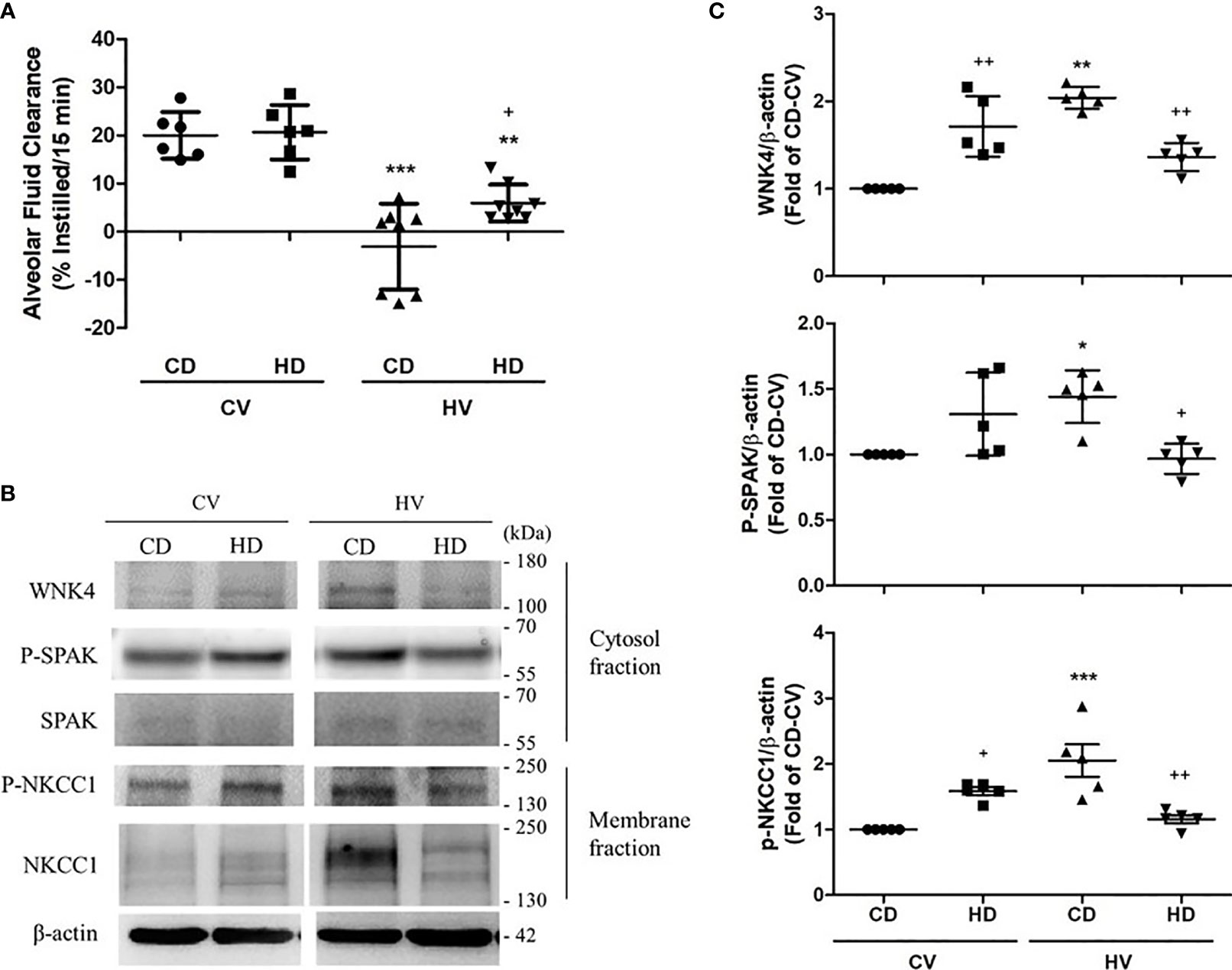 
We developed a GLP-1 analog supaglutide by fusing a pair of GLP-1 molecules with immunoglobulin constant region that contains partial hinge chains ( Kumar et al., 2007 Wang et al., 2010). In order to prolong the action of GLP-1, great efforts have been made in the past decade and two strategies are accessible: one is the use of DPP-IV inhibitors and the other is the development of DPPIV-resistant analog through structural modifications which prevent DPP-IV degradation ( Drucker et al., 2010, 2011 Lindamood and Taylor, 2015 Lee, 2016 Rajeev and Wilding, 2016). However, native GLP-1 is not suitable for therapeutic use due to its short-circulating half-life ( t1/2 < 2 min), which results mainly from rapid enzymatic inactivation by dipeptidyl peptidase-IV (DPP-IV), and/or rapid kidney clearance ( Kieffer et al., 1995). The physiological properties of GLP-1 render it as an ideal therapy for obesity and type 2 diabetes (T2D) ( Rajeev and Wilding, 2016). It exerts numerous biological functions including stimulating glucose-dependent insulin secretion, inhibiting glucagon release, inducing satiety and slowing gastric emptying ( Rajeev and Wilding, 2016). Glucagon-like peptide-1 (GLP-1) is an incretin hormone secreted by gastrointestinal L cells in response to nutrient ingestion Meier and Nauck (2005). Our findings suggest that supaglutide exerts beneficial effect on established obesity through reducing energy intake and is associated with brown remodeling of white adipose tissue. Furthermore, supaglutide improved glucose tolerance, and insulin sensitivity in the obese mice suggesting improved glucose and energy homeostasis. This is consistent with the observation that supaglutide increased tolerance of the mice to cold environment associated with up-regulation of Ucp1 in the inguinal fat. Remarkably, supaglutide reduced hepatic lipid accumulation and altered morphometry in favor of small adipocytes in fat. Importantly, the intervention significantly reduced fatty liver, decreased liver triglyceride content, and concomitantly ameliorated liver injury exemplified by declined hepatic alanine aminotransferase (ALT) and aspartic transaminase (AST) content. Compared to the placebo control, supaglutide treatment improved lipid profile, i.e., significantly decreased circulating total cholesterol levels, declined serum triglyceride, and free fatty acid levels. We found that supaglutide treatment reduced body weight, which was associated with reduced food intake. Expression of uncoupling protein 1(Ucp1) in adipose tissues was evaluated by Western blotting. Liver and adipose tissues were collected for histology analysis. Glucose tolerance and insulin resistance were evaluated by intraperitoneal glucose tolerance test and insulin tolerance tests. Cold tolerance test was performed to evaluate brown-adipose tissue (BAT) activities in response to cold challenge. Metabolic conditions were monitored and energy expenditure was assessed by indirect calorimetry (CLAMS). Mice were fed with high-fat diet (HFD) for 6 months to induce obesity and then subjected to supaglutide treatment (300 μg/kg, bi-weekly for 4 weeks), and placebo as control. Here, we investigated the effects of supaglutide in regulating energy homeostasis in obese mice. We demonstrated that this formulation possessed long-lasting GLP-1 actions and was effective in glycemic control in both type 1 and type 2 diabetes rodent models. We recently developed a novel GLP-1 mimetics (supaglutide) by genetically engineering recombinant fusion protein production techniques. However, the therapeutic use of native GLP-1 is limited due to its short half-life. GLP-1, an important incretin hormone plays an important role in the regulation of glucose homeostasis. 4Departments of Physiology and Medicine, Faculty of Medicine, University of Toronto, ON, Canada.3Division of Endocrinology and Metabolism, Keenan Research Centre for Biomedical Science, St.1Department of Endocrinology and Metabolism, Huashan Hospital, Fudan University, Shanghai, China.Yun Wan 1 Xi Bao 2 Jiabao Huang 2 Xiangyu Zhang 2 Wenjuan Liu 1,3 Qiaoli Cui 1 Dongdong Jiang 1 Zhihong Wang 1 Rui Liu 1 Qinghua Wang 1,2,3,4 *
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
